Get Started
Important Information
Get Started
Important Information
![iStock-506006436 [Converted] Home 1](https://familyfertility.com/wp-content/uploads/2024/03/iStock-506006436-Converted.png)
Welcome to the
Family Fertility Center
Having difficulty to conceive is stressful enough. Finding the right place to get help doesn’t have to be. What constitutes the “right place” depends on what matters most to you. We are an established, successful practice with distinctive credentials and attention to detail. We work with a compassionate, supportive staff that cares deeply for couples and individuals in a friendly, patient-centered environment. If this is what you are in search for, then you have found the right center for you.
Experience
Why Choose Us
Family Fertility Center is a private infertility practice founded in 1994 by Dr. H. Christina Lee. We bring hope and deliver success for couples and individuals previously unable to conceive. Thousands of families have been started at the Family Fertility Center. With our extensive experience and high success rates, we are proud to serve patients not just locally, but on an international level.
We primarily serve patients in the Lehigh Valley, Allentown, Bethlehem, Easton, Stroudsburg, Philadelphia, Harrisburg, Williamsport, Scranton, Wilkes-Barre, as well as the surrounding New Jersey and New York areas. We have also served patients from countries all over the world, such as England, France, Germany, the Netherlands, Egypt, Syria, Algeria, India, Pakistan, China, Colombia, Brazil, Venezuela and Mexico.

H. Christina Lee, M.D., J.D.
Our Physician
Our physician, Dr. H. Christina Lee is a board certified reproductive endocrinology and infertility specialist. She also is a board certified embryology laboratory director (E.L.D.), as well as a high complexity laboratory director (H.C.L.D.). Literally less than a handful of individuals in the country have such distinctive credentials.
She is uniquely qualified with extensive knowledge in the science of reproductive medicine, specialized skills in reproductive surgeries, and hands-on expertise in the IVF lab. Dr. Lee has a remarkable ability to connect with her patients. They often described her as caring, thorough, informative, humorous and dedicated. Click on Testimonials to read the unsolicited feedback provided by her patients.
The First IVF Lab in the Lehigh Valley
Qualification
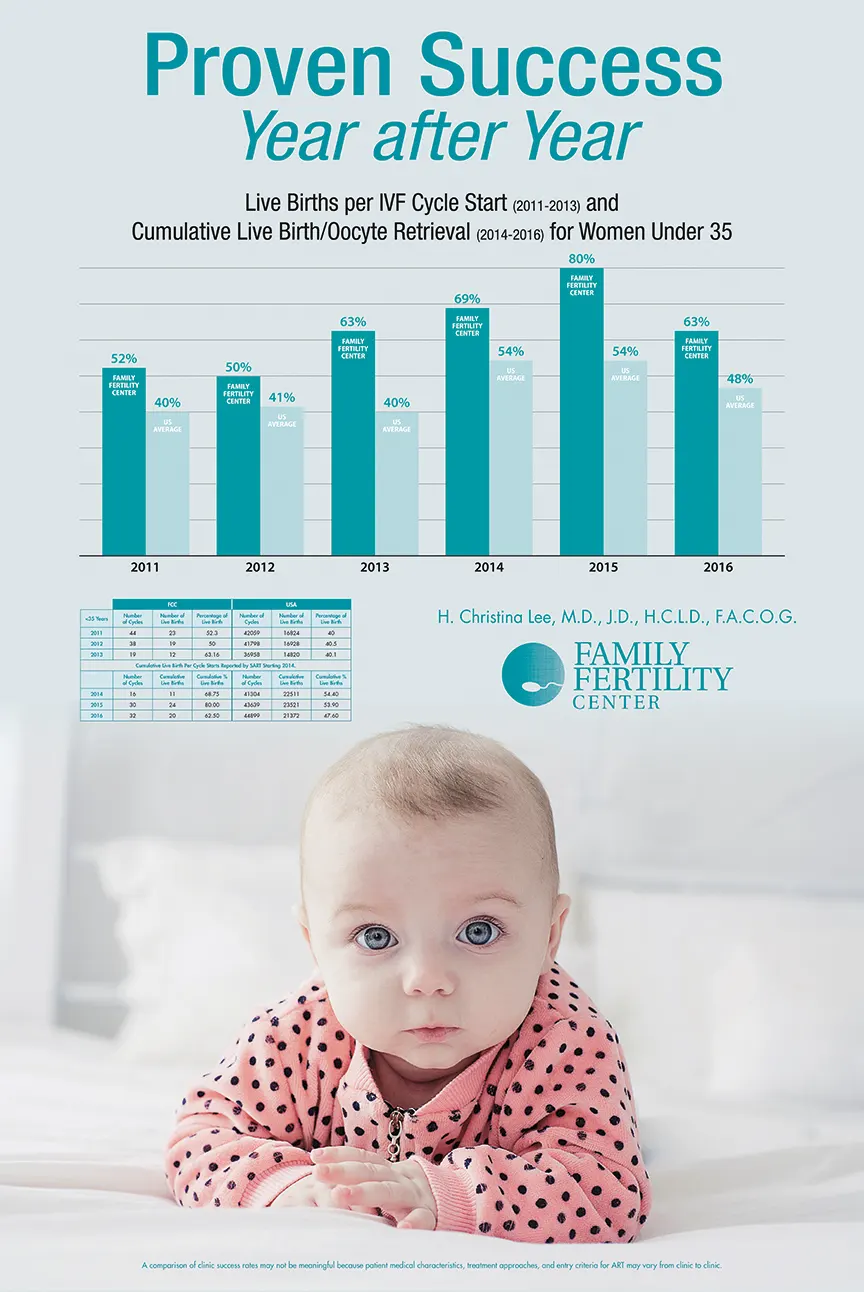
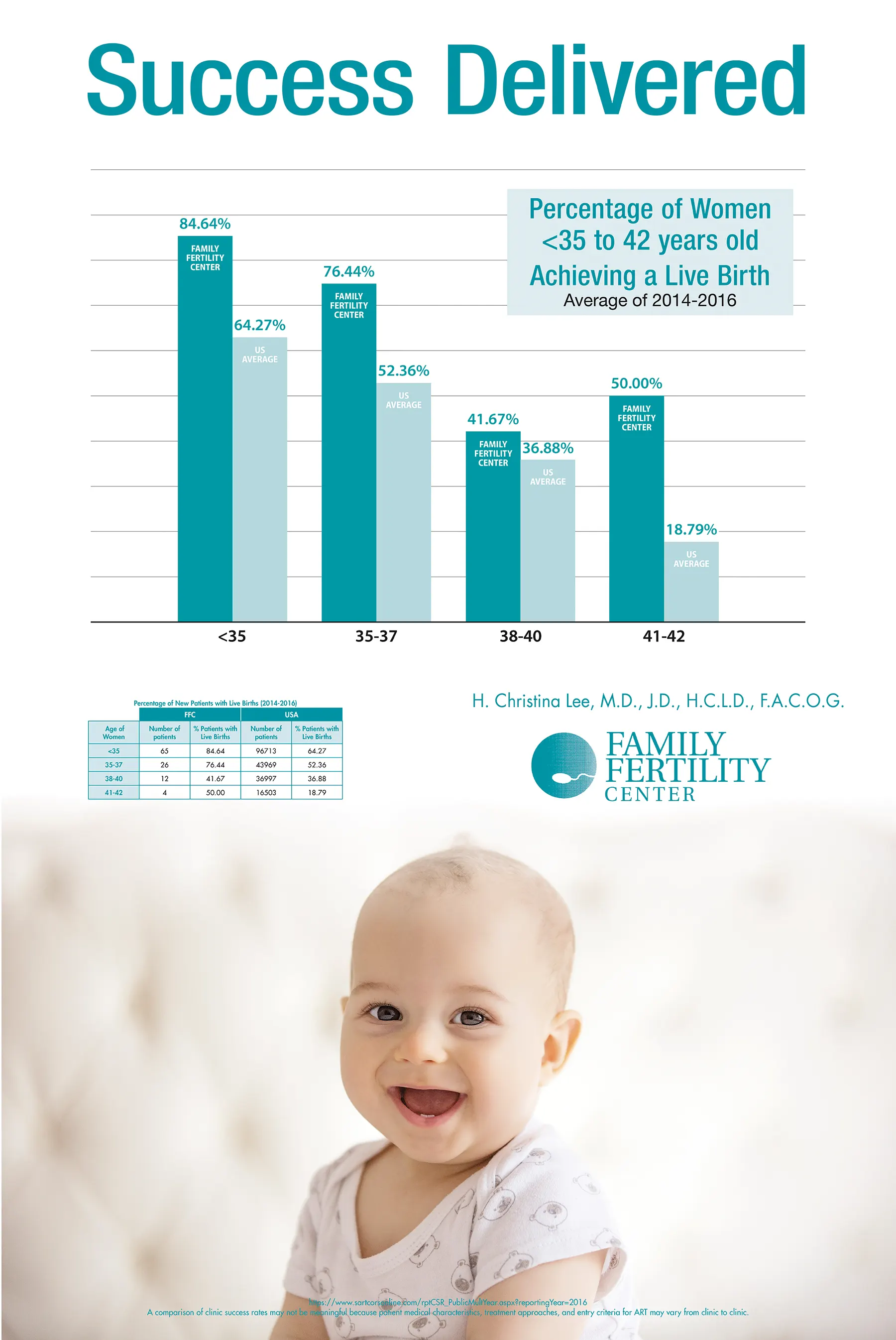
We were the first and now the only in-vitro fertilization (IVF) laboratory in the Lehigh Valley area and are fully accredited by the College of American Pathologists (CAP). Our practice is accredited by the American Institute of Ultrasound in Medicine. Our in-house hormone laboratory is Clinical Laboratory Improvement Act (C.L.I.A.) certified. And since we opened our doors in 1994, we have been a member of Society for Assisted Reproductive Technologies (S.A.R.T.), reporting success rates often surpassing the national average.
The First IVF Lab in the Lehigh Valley
Qualification
We were the first and now the only in-vitro fertilization (IVF) laboratory in the Lehigh Valley area and are fully accredited by the College of American Pathologists (CAP). Our practice is accredited by the American Institute of Ultrasound in Medicine. Our in-house hormone laboratory is Clinical Laboratory Improvement Act (C.L.I.A.) certified. And since we opened our doors in 1994, we have been a member of Society for Assisted Reproductive Technologies (S.A.R.T.), reporting success rates often surpassing the national average.
Is This Right For Me?
Fertility Specialist
A fertility specialist focuses her practice in the treatment of infertility and stays abreast of the scientific discovery and technological advances in the field. She is usually associated with a fertility center or “program”. This specialization is necessary as advanced fertility treatment often requires the fertility specialist and her staff to be available seven days a week in order to accommodate those procedures that need to be performed, even over the weekend or holiday.
Over-the-Moon in Love
Words can not express how over the moon in love we are with this little Miracle!
Thank you for making all of this possible!
Blessed to Have Found You
Just wanted to say thank you for everything – Because of you, I am a mother of two beautiful children… We are so blessed to have found you and owe all of our happiness as parents to you!
Making Our Dream a Reality
Thank you from the bottom of our hearts for making our dream of becomming parents, a reality. He has filled our lives with so much love and joy and we couldn’t be happier with our healthy little boy!
Schedule a Consultation
Contact Us
If you are experiencing a medical emergency, call 911 or go to the nearest emergency room.
We understand you may have a lot of questions. Additionally, each couple or individual has a unique set of circumstances. To this end, the best way to get answers for your situation is a face-to-face consultation with our physician.
Schedule a Consultation
Contact Us
If you are experiencing a medical emergency, call 911 or go to the nearest emergency room.
We understand you may have a lot of questions. Additionally, each couple or individual has a unique set of circumstances. To this end, the best way to get answers for your situation is a face-to-face consultation with our physician.